Endometrial Imaging – Part II
Pierre Vassallo
I n the last article, the changes observed in the appearance of the normal endometrium during the prepubertal, menarchial and reproductive periods were discussed. The current article will present the findings seen in the peri- and postmenopausal endometrium.
A strict determination of menopausal status is difficult, as the process represents a continuum that begins with perimenopause, a time span in which the patient experiences menstrual cycles of variable length and volume of flow, often with skipped cycles. Patients are officially deemed postmenopausal when they have experienced amenorrhea for 12 consecutive months from the date of the last menstrual flow, although sporadic ovulatory cycles have been reported rarely in postmenopausal women.
Although transabdominal US performed with the full bladder technique allows diagnostic imaging in some peri- and postmenopausal women, the modality has limitations. Transabdominal pelvic US may be compromised by a variety of senescent changes, including decreased urinary bladder capacity, increased body habitus, and an atrophic and illdefined endometrial canal. In many postmenopausal women, endovaginal US is required to adequately image the endometrium.
The most rapid decline in uterine size occurs within the first 10 years after menopause, with a more gradual decline thereafter. The uterus ranges from 3.5 to 7.5 cm in length and from 1.2 to 3.3 cm in anteroposterior diameter in patients older than 65 years with the wide ranges reflecting variations in patient parity, the number of years since the onset of menopause, and the presence of pre-existent uterine disease such as adenomyosis and myomas.
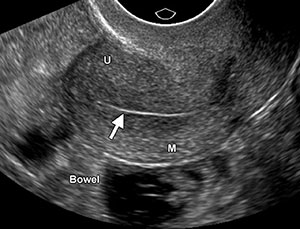
There is also a decrease in endometrial thickness from the perito the postmenopausal periods. The postmenopausal endometrium is typically atrophic because of the lack of estrogen stimulation and appears as a thin hyperechoic line or band measuring only 1–2 mm in maximum diameter (Fig 1). The relatively vascular and compact inner myometrial layer which surrounds the endometrium appears hypoechoic. A small amount of endometrial fluid, less than 2 mm in diameter, may be seen in a postmenopausal patient with an otherwise normal uterus, usually as a result of mild cervical stenosis. This fluid should be excluded from the endometrial thickness measurement (Fig 2).
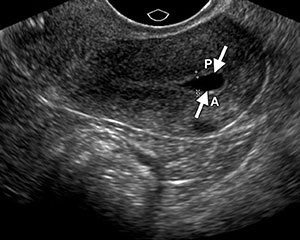
The myometrial appearance is often more heterogeneous in postmenopausal women than in premenopausal women, and the uterine body-to-cervix length ratio postmenopause approaches 1:1. Calcified uterine arcuate vessels are commonly noted in elderly postmenopausal women, especially those with diabetes, vascular disease, hypertension, or hypercalcemia (Fig 3). Free peritoneal fluid is occasionally seen in postmenopausal women and is not necessarily pathologic when small in amount and simple in appearance especially in early menopause. Free fluid in late menopause is abnormal and could be related to a variety of conditions, including intraperitoneal malignancy and ascites due to cardiac, renal, and liver disease.
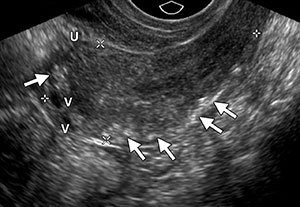
A normal endovaginal sonogram in a postmenopausal patient has a high negative predictive value and high overall accuracy rate for excluding major disorders of the uterus and ovaries, with excellent pathologic correlation. It has been shown that measurements of the endometrium obtained with endovaginal US are within 1 mm of those obtained at pathologic examination and that regardless of symptoms, an endometrial thickness of 5 mm or less is associated with benign histopathologic findings in most cases. This threshold serves as a useful guide for determining which patients should undergo endometrial biopsy. The risk of missing an endometrial abnormality with use of a single measurement of 5 mm or less at endovaginal US is low, with a 99% negative predictive value for endometrial cancer. Therefore, postmenopausal patients who present with vaginal bleeding and have an endometrial thickness of 4–5 mm or greater should be referred for further evaluation to exclude endometrial carcinoma. In postmenopausal patients without abnormal bleeding, most experts advocate tissue sampling only when the endometrial thickness exceeds 8 mm, as the risk of clinically significant endometrial abnormalities in these patients is low. In postmenopausal patients without vaginal bleeding and with an endometrial thickness from 5 to 8 mm, treatment should be based on individual symptoms and risk factors. Because endovaginal US can be used to accurately assess the endometrium, sequential US examinations can be of value for close monitoring of endometrial thickness.
Hormone replacement therapy affects the thickness of the endometrium. Sequential estrogen and progesterone therapy, often used in perimenopausal patients, induces cyclical endometrial changes and symptoms similar to those occurring in premenopausal patients. In such cases, US scans should be obtained early in the cycle or near the end of withdrawal bleeding, when the endometrium should be at its thinnest. The use of continuous estrogen and progesterone regimens leads to endometrial atrophy; therefore, measurements of endometrial thickness in these patients are usually within the normal range. Unopposed estrogen therapy is associated with an increased risk of endometrial hyperplasia or carcinoma and is typically prescribed only for women after hysterectomy.
Tamoxifen has been proven effective for the treatment of breast cancer but is associated with increased incidence of endometrial disease among postmenopausal women because of its proestrogenic effect on the uterus. Several studies have shown a direct correlation between the use of tamoxifen and an increase in endometrial thickness, most notable beyond 3 years of therapy. Nearly half of all postmenopausal women undergoing tamoxifen therapy have an endometrial thickness greater than 8 mm, and most are asymptomatic, without abnormal vaginal bleeding. The most common histopathologic findings, including hyperplasia and polyps, are benign, with an approximate 1% risk of endometrial carcinoma. Because endometrial thickening is commonly due to benign causes in patients receiving tamoxifen therapy, there is no clear consensus about a threshold thickness for recommending endometrial sampling in asymptomatic women. The generally accepted rule is that an endometrium 10mm abnormal, the latter requiring endometrial biopsy even in the absence of vaginal bleeding. However, endometrial biopsy is also performed in those patients on tamoxifen therapy who develop vaginal bleeding and have an endometrial thickness>5mm.
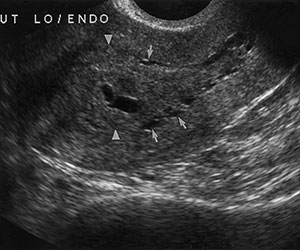
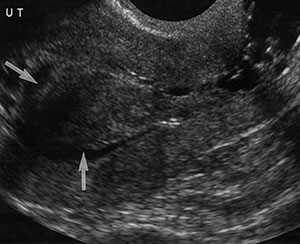
Tamoxifen causes the endometrium to appear thickened, irregular, and cystic at US (Fig 4). The punctate cystic spaces may be secondary to reactivation of adenomyosis within the inner myometrium or to obstructed glands in the endometrium due to the drug’s weak estrogenic effects. It has also been reported that the degree of endometrial thickening corresponds to the duration of tamoxifen therapy.
One of the most common reasons to perform pelvic US is postmenopausal bleeding. Causes of postmenopausal bleeding include endometrial atrophy (approximately 75% of cases), endometrial polyps, submucosal fibroids, endometrial hyperplasia, endometrial carcinoma (approximately 10%), and estrogen withdrawal. Imaging should take place immediately after bleeding has stopped, when the endometrium is presumed to be thinnest and any disease entity will be most prominent. Endometrial thickness less than 4–5 mm at endovaginal US generally excludes cancer. Any thickness greater than 5 mm in the setting of postmenopausal bleeding or any endometrial heterogeneity or focal thickening seen at endovaginal US should be investigated further with sonohysterography, biopsy, or hysteroscopy.
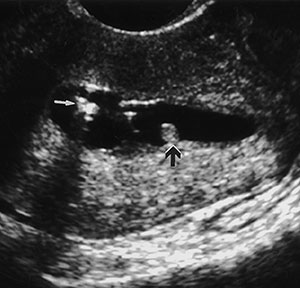
Endometrial polyps are a common cause of postmenopausal bleeding and are most frequently seen in patients receiving tamoxifen. Although endometrial polyps may be visualized at endovaginal US as nonspecific endometrial thickening, they are frequently identified as focal masses within the endometrial canal. Polyps are best seen at sonohysterography and appear as echogenic, smooth, intracavitary masses outlined by fluid (Fig 5).
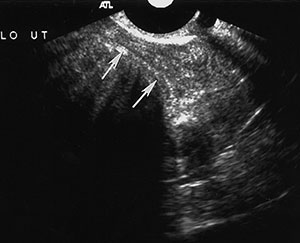
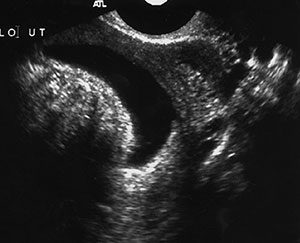
Uterine fibroids (or leiomyomas) are benign myometrial tumors that occur in patients of all ages. Although their size and frequency increase with age, they may grow until menopause and then involute and are a cause of premenopausal uterine bleeding. They are commonly identified at US as hypoechoic solid masses, but they may be heterogeneous or hyperechoic, depending on the degree of degeneration and calcification. Fibroids may interrupt the endometrium if found in a submucosal location. They are best visualized at sonohysterography (Fig 6) or hysterosalpingography
Endometrial hyperplasia is an abnormal proliferation of endometrial stroma and glands and represents a spectrum of endometrial changes ranging from glandular atypia to frank neoplasia. A definitive diagnosis can be made only with biopsy, and imaging cannot reliably allow differentiation between hyperplasia and carcinoma. Up to one-third of endometrial carcinoma is believed to be preceded by hyperplasia.
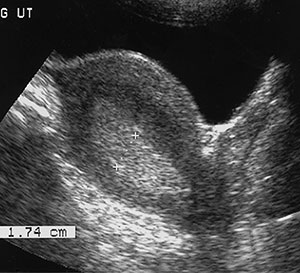
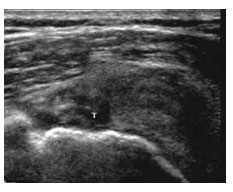
All types of endometrial hyperplasia (cystic, adenomatous, atypical) can cause diffusely smooth or, less commonly, focal hyperechoic endometrial thickening (Fig 7). The US appearance can simulate that of normal thickening during the secretory phase, sessile polyps, submucosal fibroids, cancer, and adherent blood clots. Endometrial hyperplasia is considered whenever the endometrium appears to exceed 10 mm in thickness, especially in menopausal patients, although it can be reliably excluded in these patients only when the endometrium measures less than 6 mm.
Endometrial hyperplasia may also cause asymmetric thickening with surface irregularity, an appearance that is suspicious for carcinoma. Because endometrial hyperplasia has a nonspecific appearance, any focal abnormality should lead to biopsy if there is clinical suspicion for malignancy.
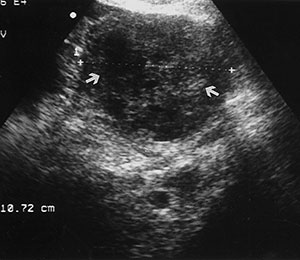
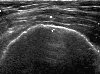
Endometrial adenocarcinoma is the most common invasive gynaecologic malignancy, but thanks to early detection and treatment, it is not a leading cause of cancer death. US signs of endometrial carcinoma include heterogeneity and irregular endometrial thickening (Fig 8). These signs are nonspecific and can be seen in endometrial hyperplasia as well as polyps, leading to biopsy of almost any irregularity in the setting of postmenopausal bleeding. However, polypoid tumors tend to cause more diffuse and irregular thickening than a polyp and more heterogeneity than endometrial hyperplasia. A more specific US sign is irregularity of the endometrium-myometrium border, a finding that indicates invasive disease. A small amount of fluid in the endometrial canal is likely to be related to benign cervical stenosis and does not require further evaluation. A large intrauterine fluid collection in a postmenopausal patient, although possibly related to cervical stenosis, should raise concern for endometrial (or cervical) carcinoma.
This article outlines the normal findings seen on ultrasound of the endometrium in peri and post menopausal women and concludes by identifying the most commonly encountered disease entities and their sonographic features.
 |
 |
| Fig. 6a | Fig. 6b |
 |
 |
| Fig. 7 | Fig. 8 |