Management of consumers’ pharmaceutical waste in a pharmacy setting – Part II
by Karen Attard, Maurice ZarbAdami, Lilian M. Azzopardi and Anthony Serracino-Inglott
Incompatible products and liquid medicines
Products that might cause particular hazards if mixed should be kept separately in order to protect from consequences of mixing incompatible products. Thus, liquids, syrups, suspensions, inhalers, creams, ointments will all have to be returned directly to the pharmacist in their original containers and stored in a metal cabinet until the date of collection by a waste carrier.1
Waste containers
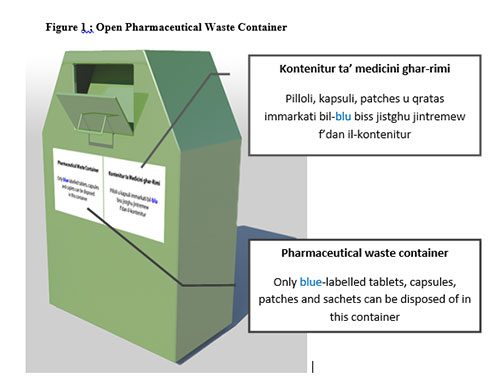
As part of this scheme, consumers will receive durable re-sealable plastic bags to place their pharmaceutical waste. Special waste containers (Figure 1) will be placed in front of pharmacies. They should be made of a metal resistant to corrosion and fire and less prone to theft. The colour chosen (in this case green) should be such that makes the container distinguishable from post boxes and garbage bins. Once the lid is opened, the pharmaceutical waste can be placed and it cannot be retrieved again. Labels will clearly show the purpose of this container and it will instruct what can be disposed of in this manner and what cannot be disposed as such.
The Returned Medicines Register
The ‘Returned Medicines Register’ (Table 1) is used to keep a record of all the medicines returned directly to the pharmacy. It should be in the form of carbon copy paper so as to give a receipt to the Waste collector upon collection of the waste.2 The Returned Medicines Register should be concise but still containing all the relevant information needed upon collection, while not very time-consuming.
Pharmaceutical waste management
It is imperative to distinguish between hazardous medicines and hazardous pharmaceutical waste. They will all be ultimately incinerated, but it is important for them to be segregated at the point of collection.
Hazardous medicines
Hazardous medicines should be returned to the pharmacist by hand. The returned medicines should be left in their original packaging and they should be placed in the specialised sealable bags.
Once returned to the pharmacist, they should be stored in a lockable metal cabinet and logged on the ‘Returned Medicine Register’.
Non-hazardous solid dosage forms
Non-hazardous solid dosage forms such as tablets, capsules, caplets, patches and powders (sachets) should be disposed of in their original packaging in the waste container found in front of the pharmacies (Figure 1). Any quantity may be discarded in this manner provided that they are still in their original packaging and they are not hazardous in nature.
Non-hazardous non-solid dosage forms
This refers to syrups, suspensions, creams, ointments and inhalers. These should be left in their original packaging and placed in the special sealable bags before returned to the pharmacist. However these are not disposed of in the waste container to minimise the risk of contamination and mixing of incompatible products but are returned by hand to the pharmacist. Again, all the returns have to be logged on the ‘Returned Medicine Register’.
Waste management in the pharmacy
A system should be in place so that each pharmacy should manage its own pharmaceutical waste by returning them to the respective agents. The agents should then contract a private waste management company to manage their waste in an ethical and safe way ensuring total compliance with legislation.
Facilitating the segregation process
In order to facilitate the segregation between hazardous waste ie waste which should be disposed of in the waste container and waste which should be returned to the pharmacist; each medicinal product should be colour coded respectively with small coloured stickers. This can be done either by the manufacturers, by the importers or by staff at the pharmacy during pricing. The colour codes which can be used include:
RED – for hazardous medicines
BLUE – for waste which can be disposed of in the waste containers
GREEN – for waste which should be returned directly to the pharmacist
This is done to help people use the proposed scheme successfully and to minimise errors.
Waste carriers
Waste carriers will be contracted by waste contractors to visit the pharmacies weekly. The waste container is emptied once weekly and the pharmaceutical waste should be collected together with a copy of the register, so as to know what kind of waste is present to reduce hazards when handled, and to allow for eventual safe incineration.
Waste contractors
Waste contractors are companies specialised in the field of waste management. These obtain the necessary permits from local authorities and manage waste of different origin according to European standards. They are in charge of:
- Collecting waste;
- Transporting waste in authorised appropriate vehicles;
- Storing waste in specialised stores when required;
- Ethically and safely managing and disposing of waste as required by European standards.
Best management practice
The best management practice is High Temperature Incineration (HTI), carried out in a licensed and documented manner to provide a full audit trail.3 Since Malta does not have an HTI facility which reaches the 1100°C required to incinerate hazardous pharmaceutical waste, the best solution is the exportation of such pharmaceutical waste to incineration sites in the EU for eventual complete destruction.
Acknowledgement
The authors acknowledge the assistance of Mr Clayton Curmi and Mr Nicholas Caruana with the design of the waste containers.
References
- Health Technical Memorandum 07-06: disposal of pharmaceutical waste in community pharmacies. NHS Estates; 2007
- CRG Medical foundation for patient safety. U.S. National Strategy for unused or expired medications questionnaire for returned medication by individual donors, 2005.
- Directive 2000/76/EC on the incineration of waste. Available from: http://europa.eu/legislation_summaries/environment/waste_management/l28072_en.htm




