Valsartan: Updated list of potentially impacted products
Medical News Archived
The Superintendent of Public Health has issued an updated list of products with the active substance valsartan that may be potentially impacted by the N-nitrosodimethylamine (NDMA) impurity.

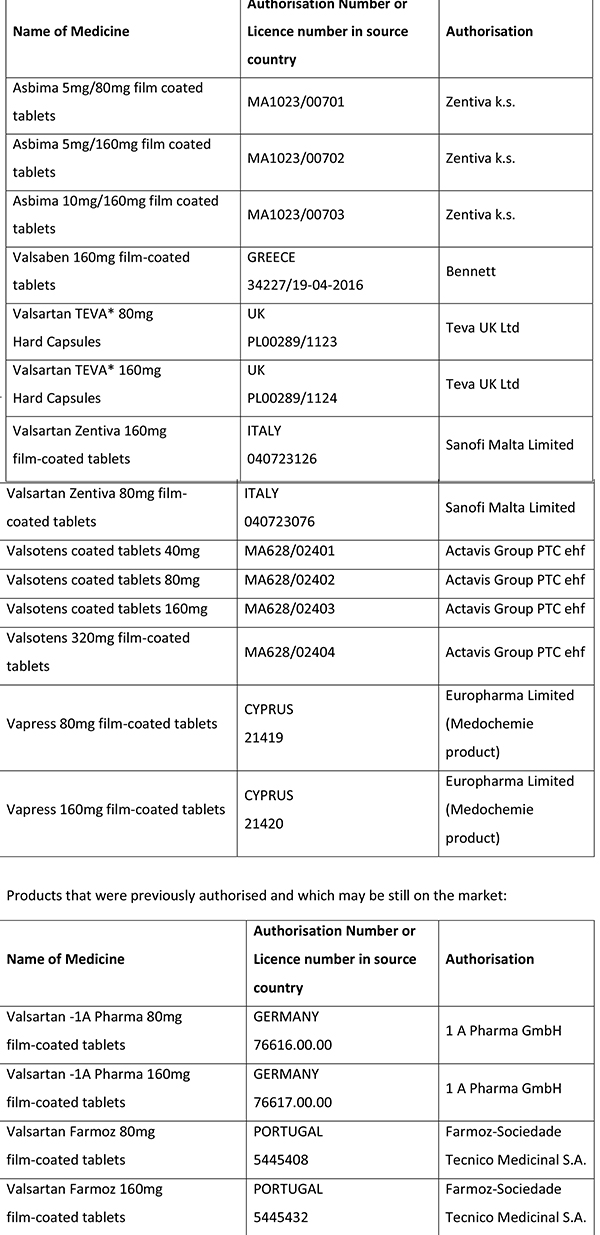
It is important to note that treatment should not be stopped until the patient has been prescribed an alternative. The health risk of discontinuing this medicine is higher than any potential risk presented by the impurity. At present there is no evidence that the impurity in some stocks of valsartan has caused any harm to patients. Further updates will be provided as more information becomes available.
Tags
adoloscence
cancer
cannabis
Cardiology
Contributions
covid 19
dementia
Dermatology
Dermfest
Diabetes
Editorial
endocrinology
environmental health
exercise
Family Medicine
Francesco Carelli
gastroenterology
Geriatrics
gynaecology
hypertension
Infectious Diseases
Medical Education
Medical Ethics
Medicine and the arts
microbionta
Neurology
Nutrition
obesity
obstetrics
Oncology
Orthopaedics
Paediatrics
Pharmacology
Pharmacy Practice
pregnancy
Psychiatry
Public Health
Radiology
Respiratory Medicine
Rheumatology
smoking
Surgery
TheSynapse Interview
Urology
Women's health