Colorectal cancer
by Pierre Vassallo
MD PhD FACA Artz fur Radiologie
Consultant Radiologist
Colorectal cancer is the third most common cancer and the second most common cause of cancer deaths in the western world. It occurs with equal frequency in men and women. Adenomatous polyps are the known precursors of the majority of colorectal cancers, with the risk of malignancy increasing with increasing polyp size. Detection of these polyps followed by polypectomy has been found to prevent the development of colorectal carcinoma. Despite the screening test options currently available, the majority of people who should undergo screening for colorectal cancer do not do so.
Four screening tests are routinely used for the detection of colorectal cancer. Current colorectal cancer screening options include faecal occult blood testing, flexible sigmoidoscopy, air-contrast barium enema examination and fiberoptic colonoscopy, whilst a more recently introduced test consists of CT colonography.
The faecal occult blood test is safe and inexpensive. However, its performance is poor, since most colon cancers bleed intermittently and most adenomatous polyps do not bleed. The sensitivity of the faecal occult blood test as a single test for colorectal cancer is 20%–30% and for a large polyp is 10%–15%. The sensitivity of this test for colorectal cancer increases with repeated screening and ranges between 72% and 78%. Many causes of false-positive results exist, such as upper gastrointestinal tract sources of bleeding. However, randomised controlled trials have shown that the faecal occult blood test confers a 15%–33% mortality reduction from colon cancer.
Flexible sigmoidoscopy allows examination of about 60 cm of the colon, and therefore only 40%–65% of lesions are within reach of the sigmoidoscope. Up to 50% of proximal cancers are not associated with a distal index polyp. A recent study in 2,885 patients showed that a combination of faecal occult blood testing and flexible sigmoidoscopy failed to detect 24% of cases of advanced colorectal cancer. However, periodic sigmoidoscopy has been shown to reduce colorectal cancer mortality by 60%–80%.
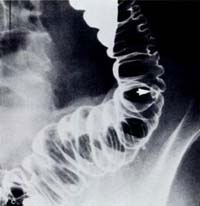
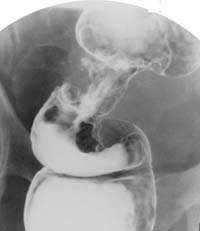
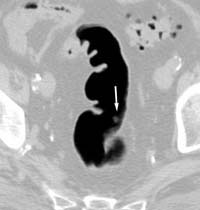
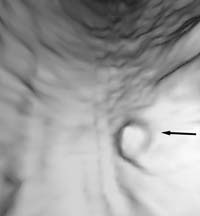
Double-contrast barium enema examination has been found to have sensitivities ranging from 71% to 95% in the detection of colon carcinoma (Figure 1) in retrospective studies and is generally considered to be an excellent test for the detection of clinically significant lesions. However, the sensitivity decreases with polyps (Figure 2) and adenomas smaller than 10mm in diameter.
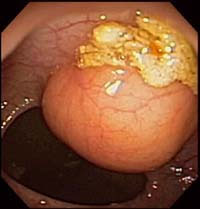
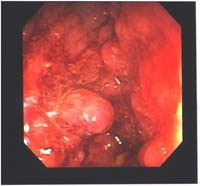
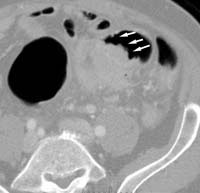
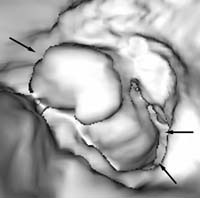
Fiberoptic colonoscopy is considered the gold standard for colon evaluation (Figures 3 and 4). However, colonoscopy has also been found to miss polyps with a miss rate of 24% for adenomas overall, 27% for adenomas 5 mm in diameter or smaller, 13% for adenomas 6–9 mm in diameter, and 6% for adenomas 10 mm in diameter or larger. Other limitations of colonoscopy include extremely variable patient compliance, the need for multidrug intravenous sedation, and the high cost of the test. In addition, the procedure is time-consuming and is incomplete in up to 10% of cases due to colonic anatomy (long or very tortuous colon).
CT colonography was first introduced in 1994 and has received widespread attention as a possible screening tool for colorectal polyps and cancer that may help to increase the rate of patient compliance. CT colonography involves the use of spiral CT data in combination with advanced graphical software to generate two-dimensional views and three-dimensional endoluminal views of the colon (Figure 5), which may be viewed dynamically and interactively on a computer workstation to simulate a conventional colonoscopy. Adequate colonic preparation and distension are necessary to ensure maximal diagnostic quality and patients must still undergo a colonic cleansing regimen before CT colonography. CT colonography and conventional colonoscopy have been shown to have equivalent sensitivity in the detection of the 10-mm-diameter or larger polyps that are considered to be clinically significant. CT colonography has been found useful in the evaluation of the colon proximal to a lesion that is causing distal obstruction (Figure 6) and in the setting of failed colonoscopy. Several published studies have documented the ability of CT colonography to show the cause of obstruction, as well as to depict additional cancers and polyps in the colon proximal to the distal lesion.
The advantages of CT colonography are that it presents minimal risk to patients, has a short procedure time (about 10 minutes), can be performed in patients with distal occluding lesions, and can be used to localize lesions more precisely than with colonoscopy. CT colonography also allows diagnosis of extracolonic findings and screening for other clinically important diseases. Moderately significant findings such as gallstones, as well as highly significant findings such as renal cell carcinoma, large abdominal aortic aneurysms, and liver and adrenal masses can be identified.
CT colonography is limited, in that patients must still undergo bowel cleansing. Poor preparation or poor distension will compromise the diagnostic ability of the CT and small and flat polyps are not well detected. Also conventional colonoscopy allows direct biopsy of visualised lesions, while lesions detected with CT colonography require conventional colonoscopic biopsy.
Future developments include stool DNA testing which may be incorporated into colorectal cancer screening algorithms. Colonic neoplasms continuously exfoliate various DNA markers. Targeting multiple DNA mutations found in stool samples may achieve high rates of neoplasm detection with a sensitivity of 91% for cancer and 82% for large adenomas. Mutations on K-ras, APC, and p53 genes, as well as mutations on Bat-26 (a microsatellite instability marker) and long DNA have all been found to be markers for colorectal carcinoma.
The American Cancer Society recommendations for colon cancer screening in average-risk adults 50 years of age and older include:
- annual faecal occult blood testing, or
- flexible sigmoidoscopy every 5 years, or
- annual faecal occult blood testing and flexible sigmoidoscopy every 5 years, or
- double-contrast barium enema examination every 5 years, or
- colonoscopy every 10 years.
For patients with higher risk, screening is performed according to Table A.
Table A
| Risk Category | Age to Begin | Recommendation | Comments |
| INCREASED RISK | |||
| Previous single, small (< 1 cm) adenomas | Within 3 years | Colonoscopy | If normal, screen as average risk |
| Previous single large (>1 cm ) or multiple adenomas, or high-grade dysplasia or villous change. | Within 3 years | Colonoscopy | If normal, repeat in 5 years; If again normal screen as average risk |
| Previous curative-intent resection of colorectal cancer | Within 1 year | Colonoscopy | If normal, repeat in 5 years; If again normal, repeat every 5 years |
| Colorectal cancer or adenomatous polyps, in first-degree relative before age 50, or more first-degree relatives at any age if no hereditary syndrome | Age 40, or 10 years before the youngest case in the immediate family | Colonoscopy | Every 5-10 years. Colorectal cancer in relatives more distant than first-degree does not increase risk substantially above the average risk group. |
| HIGH RISK | |||
| Family history of familial adenomatous polyposis (FAP) | Puberty | Early surveillance with colonoscopy, and consider genetic testing | If the genetic test is positive, colectomy is indicated* |
| Family history of hereditary non-polyposis colon cancer (HNPCC) | Age 21 | Colonoscopy and consider genetic testing | If the genetic test is positive or if the patient has not had genetic testing, every 1-2 years until age 40, then annually* |
| Inflammatory bowel disease: Chronic ulcerative colitis, Crohn’s disease | 8 yrs after onset of pancolitis, or 12yrs after onset of left-sided colitis | Colonoscopy with biopsies for dysplasia | Every 1-2 years* |
*These patients are best referred to a centre with experience in the surveillance and management of FAP, HNPCC or IBD.