UPDATE Information regarding Valsartan containing products
The situation regarding valsartan containing products that may be effected has been issued by the Medicines Authority Malta
The release is available on http://www.medicinesauthority.gov.mt/news-details?id=FF9F and is being reproduced below

Obviously this is an evolving situation and more details will be forwarded as soon as they are officially communicated to us.
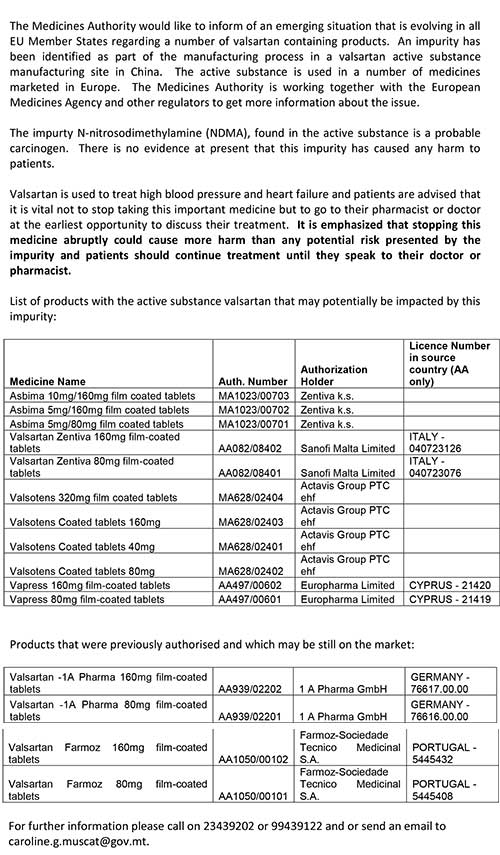
Letter from the Superintendent of Public Health available from here: http://thesynapse.net/images/articles/Valsartan_Doctor.pdf
Other useful links are the following:
http://www.hpra.ie/docs/default-source/Safety-Notices/valsartan/final-valsartan-recall-letter-05-07-2018.pdf?sfvrsn=2 – This contains a detailed list of the relevant companies affected in the Republic of Ireland
EMEA Press release issued 5 July 2018 – http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2018/07/news_detail_002984.jsp&mid=WC0b01ac058004d5c1
EMEA News and events page – http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/landing/news_and_events.jsp&mid=
MHRA News re recall https://assets.publishing.service.gov.uk/media/5b3e325de5274a37707745fa/EL_18_A_11.pdf