Management of consumers’ pharmaceutical waste in a pharmacy setting
by Karen Attard, Maurice ZarbAdami, Lilian M. Azzopardi & Anthony Serracino-Inglott
The management of pharmaceutical waste is increasingly becoming of great concern to our society since no official program currently exists. This study proposes a management plan for consumer’s pharmaceutical waste in a pharmacy setting in Malta. It involves disposing of unwanted solid dosage-forms in their original packaging in waste containers which will be installed in front of pharmacies, and returning hazardous medicines and other dosage-forms directly to the pharmacist to reduce hazards from mixing incompatible products.
Key words
Pharmaceutical waste management; pharmacy; consumers; waste containers.
Introduction
Ever since joining the European Union, Malta has been trying extensively to conform to European standards as requested by legislation. Improvements have been registered in several sectors; however the research in the management of waste pharmaceuticals has so far remained lackinge.[1]
Aims
This report draws findings of a scientific study that has been undertaken by the authors and aims to provide a programme and a guidance proposal plan for the management and disposal of pharmaceutical waste produced by consumers. The process of designing and implementing a successful pharmaceutical waste management program, being highly interdisciplinary, has to be acceptable for pharmacies, competent authorities and consumers alike.[2]
Educational programs
Educational programs should be set up to educate consumers on the threats resulting from the irresponsible disposal of pharmaceutical waste. The proposed programs should educate users on new, well thought-out and science-founded procedures.[3] The benefits should be highlighted to encourage people to make an effort to abide by these guidelines.
Such educational programs include:
- Promoting the campaign on television programs including waste management ‘spots’ on TV;
- Advertisements on published media;
- Distribution of leaflets from community pharmacies;
- Other advertising material including banners and posters.
Consumers’ pharmaceutical waste
Examples of consumers’ pharmaceutical waste include:
- Expired medicines in households;
- Medicines discontinued by manufacturers due to a negative risk-benefit ratio;
- Medicines not consumed by patients;
- Medicines discontinued because the doctor prescribed new medication;
- Medicines of deceased patients.
Since pharmaceutical waste production in nursing homes is significantly higher than in households, this scheme is not applicable. In this setting, clinical waste is also produced and thus nursing homes should contract a private waste management company to manage their pharmaceutical and clinical waste. Hospitals have their in-house waste management procedures.
Household waste medicines are found in various dosage forms; the most common being tablets, capsules, caplets, syrups, suspensions, inhalers, suppositories, pessaries, creams, ointments, sprays, drops, powder for oral constitution and patches.[4]
Syringes are not included in this scheme. Yellow UN approved containers should be distributed to frequent syringe users from healthcare centres. When the yellow bin is full, consumers should return the bin to the centre and they are given an empty one. The bins are then collected from the healthcare centres by waste carriers.
Hazardous waste
Section 4(2) of the Waste Management Act of 2001 defines Hazardous Waste as having certain properties which can be harmful to human health and the environment. “These properties are corrosive, explosive, toxic, flammable, oxidizing, irritant, harmful, carcinogenic, infectious, teratogenic, mutagenic and ecotoxic.”
Medicines may have a range of hazardous properties, but only those with one of the hazardous properties denoted by “H codes” will be classified as a hazardous waste.5 Other medicines are not hazardous within the meaning of the legislation, but they are hazardous if released in the environment.
Hazardous medicines
The European Waste Catalogue5 lists wastes from human healthcare in Chapter 18. Medicines consisting of or containing dangerous substances together with cytotoxic and cytostatic medicines are considered to be hazardous. Hazardous medicines include chemotherapeutic agents, antivirals, hormones and some bioengineered medicines.[6] Most of the medicines available in community pharmacies are fairly inert and pose no particular danger to staff or waste carriers who collect the waste. This is because most medicines are individually packed tablets or capsules. The packaging provides satisfactory protection for anyone handling the container. However if the solid formulation is altered by, for example, being crushed, they could pose a risk. Blister packaging also protects against mixing incompatible products and they do not pose a risk to the environment provided that they are incinerated properly. 2
Pharmacies should develop a list of hazardous medicines that are found in the pharmacy, including medicines available from the Pharmacy of Your Choice (POYC Scheme) so as to be aware of any risks involved during collection.
Anti-acne preparations |
||
|
Active ingredient |
Trade Name |
Formulation |
|
Clindamycin 10mg/ml |
Dalacin T |
Topical solution |
|
Isotretinoin 10mg, 20mg |
Decutan |
Capsule, Soft |
Anti-bacterials for systemic use |
||
|
Clavulanic Acid 125mg; Amoxicillin 500mg |
Augmentin-Duo 500mg/125mg |
Film-Coated Tablet |
|
Cefaclor 500mg |
Ceclor |
MR tablets |
Antibiotics and chemotherapeutics for dermatological use |
||
|
Retampulin |
Altargo |
Ointment |
|
Mupirocin 2% w/w |
Bactroban |
Topical ointment |
Intestinal anti-inflammatory and anti-infective agents |
||
|
Mesalazine 400mg |
Pentacol |
Gastro Resistant Tablet |
|
Sulfasalazine 500mg |
Salazopyrin |
Suppositories |
Anti-gout preparation |
||
|
Allopurinol 100mg |
Allopurinol |
Tablet |
|
Colchicine 500mcg |
Colchicine |
Tablet |
Anti- neoplastic agents |
||
|
Methotrexate 2.5mg |
Methotrexate |
Film-Coated Tablet |
|
Imatinib 100mg |
Imatinib |
Tablets |
Endocrine therapy |
||
|
Letrozole 2.5mg |
Femara |
Tablet |
|
Tamoxifen 20mg |
Nolvadex D |
Film-Coated Tablet |
Ophtalmologicals |
||
|
Tobramycin 3mg/ml |
Tobrex |
Eye Drops, Solution |
|
Aciclovir 3% w/w |
Zovirax |
Eye Ointment |
Sex hormones and modulators of the genital system |
||
|
Estradiol 25mcg |
Vagifem |
Vaginal Tablet |
|
Ethinylestradiol 0.03mg, Drospirenone 3mg |
Yasmin |
Film-Coated Tablet |
Thyroid therapy |
||
|
Levothyroxine Sodium 100mcg |
Eltroxin |
Tablet |
|
Carbimazole 5mg |
NeoMercazole 5 |
Tablet |
Table 1 : Hazardous medicines available in pharmacies (Source: Malta Medicines List)
This table is only a summary of the full version which is part of the scientific study entitled ‘Waste Management in Pharmacy’.
Table 1 is a short template of a list of medicines available in pharmacies which are considered to be hazardous, that was compiled as part of the scientific study undertaken. This list should be modified according to what is available at each respective pharmacy.
Incompatible products and liquid medicines
Products that might cause particular hazards if mixed should be kept separately in order to protect from consequences of mixing incompatible products. Thus, liquids, syrups, suspensions, inhalers, creams, ointments will all have to be returned directly to the pharmacist in their original containers and stored in a metal cabinet until the date of collection by a waste carrier.2
Waste containers
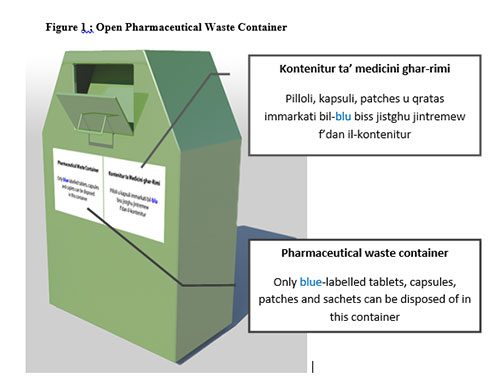
As part of this scheme, consumers will receive durable re-sealable plastic bags to place their pharmaceutical waste. Special waste containers (Figure 1) will be placed in front of pharmacies. They should be made of a metal resistant to corrosion and fire and less prone to theft. The colour chosen should be such that makes the container distinguishable from post boxes and garbage bins. Once the lid is opened, the pharmaceutical waste can be placed and it cannot be retrieved again. Labels will clearly show the purpose of this container and it will instruct what can be disposed of in this manner and what cannot be disposed as such.
The Returned Medicines Register
The ‘Returned Medicines Register’(Table 2) is used to keep a record of all the medicines returned directly to the pharmacy. It should be in the form of carbon copy paper so as to give a receipt to the Waste collector upon collection of the waste.[7] The Returned Medicines Register should be concise but still containing all the relevant information needed upon collection, while not very time-consuming.
Pharmaceutical waste management
It is imperative to distinguish between hazardous medicines and non-hazardous medicines. They will all be ultimately incinerated, but it is important for them to be segregated at the point of collection. Non- hazardous solid dosage forms such as tablets, capsules, caplets, patches and powders (sachets) should be disposed of in their original packaging in the waste container found in front of the pharmacies. Any quantity may be discarded in this manner provided that they are still in their original packaging and they are not hazardous in nature. On the other hand Non-hazardous non-solid medicines (syrups, suspensions, creams, ointments and inhalers) should also be left in their original packaging and placed in the special sealable bags before returned to the pharmacist. However these are not disposed of in the waste container to minimise the risk of contamination and mixing of incompatible products but are returned by hand to the pharmacist. Again, all the returns have to be logged on the ‘Returned Medicine Register’.
Waste management in the pharmacy
A system should be in place so that each pharmacy should manage its own pharmaceutical waste by returning them to the respective agents. The agents should then contract a private waste management company to manage their waste in an ethical and safe way ensuring total compliance with legislation.
Facilitating the segregation process
In order to facilitate the segregation between hazardous waste ie waste which should be disposed of in the waste container and waste which should be returned to the pharmacist; each medicinal product should be colour coded respectively with small coloured stickers. This can be done either by the manufacturers, by the importers or by staff at the pharmacy during pricing. The colour codes which can be used include:
RED – for hazardous medicines
BLUE – for waste which can be disposed of in the waste containers
GREEN – for waste which should be returned directly to the pharmacist
This is done to help people use the proposed scheme successfully and to minimise errors.
Waste carriers
Waste carriers will be contracted by waste contractors to visit the pharmacies weekly. The waste container is emptied once weekly and the pharmaceutical waste should be collected together with a copy of the register, so as to know what kind of waste is present to reduce hazards when handled, and to allow for eventual safe incineration.
Waste contractors
Waste contractors are companies specialised in the field of waste management. These obtain the necessary permits from local authorities and manage waste of different origin according to European standards. They are in charge of:
- Collecting waste;
- Transporting waste in authorised appropriate vehicles;
- Storing waste in specialised stores when required;
- Ethically and safely managing and disposing of waste as required by European standards.
Best management practice
The best management practice is High Temperature Incineration (HTI), carried out in a licensed and documented manner to provide a full audit trail.[8] Since the best solution is the exportation of such pharmaceutical waste to incineration sites in the EU for eventual complete destruction.
Acknowledgement
The author(s) acknowledge the assistance of Mr Clayton Curmi and Mr Nicholas Caruana with the design of the waste containers.
[1]. Muscat MA. Setting up guidelines for proper pharmaceutical waste disposal [Dissertation]. University of Malta; 2006.
[2]. Health Technical Memorandum 07-06: disposal of pharmaceutical waste in community pharmacies; 2007
[3]. Hasan SE. Public awareness is Key to successful waste management. J Environ Sci Health A 2005; 39(2):483-92.
[4]. Azzopardi LM. Lecture Notes in Pharmacy Practice. London. Pharmaceutical Press, 2010.
[5]. Environment Protection Agency. European waste catalogue and Hazardous waste list, 2002. Available from: http://www.environ.ie/en/Publications/Environment/Waste/WEEE/FileDownload,1343,en.pdf
[6]. Department of Health and Human Services. NIOSH list of antineoplastic and other hazardous drugs in healthcare setting, 2010.
[7]. CRG Medical foundation for patient safety. U.S. National Strategy for unused or expired medications questionnaire for returned medication by individual donors, 2005.
[8]. Waste Incineration Directive 2000/76/ECAvailable from: http://europa.eu/legislation_summaries/environment/waste_management/l28072_en.htm