Brugada Syndrome and Sudden Cardiac Death
Samuel Meilak & Mark Sammut
Abstract
A recently discovered cause of sudden cardiac death, Brugada syndrome is a conductivity disorder in structurally normal hearts. We present here a case of an incidental Brugada sign, outlining the clinical and pathophysiological findings in Brugada syndrome. We also give an account of the published guidelines regarding implantable cardioverter-defibrillators and highlight the importance of educating clinicians and the public alike in the management of Brugada syndrome.
Case presentation
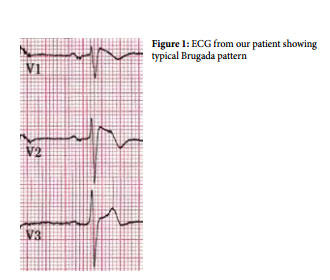
A 49 year old gentleman, with no relevant medical history, presented with epigastric discomfort associated with sweating but no nausea or vomiting. He was haemodynamically stable and clinical examination proved unremarkable. The electrocardiogram (ECG) showed ST segment elevation in the right chest leads whilst serial troponin levels were within normal range. Coronary angiogram showed normal contractility and no significant coronary artery disease. The working diagnosis was of epigastric discomfort secondary to dyspepsia, with the Brugada sign being an incidental ECG finding. The plan was for this patient to be managed expectantly and with regular ECGs.
History of Brugada syndrome
The first paper on Brugada syndrome described eight cases and was published in 1992. It describes the first documented case which occurred in a three year old Polish boy who in 1986 presented with multiple episodes of loss of consciousness; he was resuscitated repeatedly by his father. The child’s sister had died suddenly at two years of age. The ECGs of the both siblings were very similar and abnormal. In recent years there has been an exponential increase in the number of cases reported1 .
Recent genetic profiling seems to indicate that Brugada syndrome is a primary conduction disorder2 .
Brugada Syndrome
This channelopathy is characterised by sudden death associated with one or more of several ECG patterns i.e. incomplete right bundle branch block (RBBB); ST elevation; and J-point elevation in the anterior precordial leads (V1 -V3 ). Three different types have been described, classified according to their characteristic ST segment abnormalities in leads V1 – V3 3 . It is debatable whether the RBBB is real or reflecting early repolarisation of the right ventricular epicardium. In some but not all cases this resolves after normalisation of the ST segment. In addition, the PR interval is usually increased, reflecting a prolongation in the H-V interval.
There is a propensity for life-threatening ventricular arrhythmias, with the complete syndrome being characterised by episodes of fast polymorphic ventricular tachycardias (VT). These may range from self-limiting runs causing syncopal attacks to ongoing arrhythmias causing a full blown cardiac arrest and sudden cardiac death.
The above mentioned abnormalities occur in the setting of a structurally normal heart.
Pathophysiology
Recent research has implied a loss-of-function mutation of the SCN5A gene in about 10-30% of patients with Brugada syndrome. This affects the cardiac voltage-gated sodium channel (Nav 1.5) leading to a reduction in the sodium current (INa) available during the phases 0 (upstroke) and 1 (early repolarisation) of the cardiac action potential.
One of the hypotheses which have been put forward to explain the underlying mechanism is the ‘Repolarisation-defect theory’. It proposes that there is an increased contribution of the transient outward current (Ito) to the action potential waveform in right ventricular epicardial cells compared to endocardial cells (giving a more prominent notch in the action potential of the former). This difference is accentuated by a decrease in INa, creating a voltage gradient during repolarisation and the characteristic morphology of ST elevation2 . Another hypothesis explains the ECG findings as a reflection of disordered conduction (delayed activation and slow conduction) in the right ventricle, mainly the outflow tract. This is known as the depolarisation hypothesis.
Diagnosis
A patient may be completely asymptomatic and said to have the Brugada sign. Those suffering from Brugada syndrome typically present with syncope but sudden cardiac death may also occur, commonly during sleep. Physical examination is unremarkable whilst there is usually a strong family history of sudden cardiac death.
Investigations:
1. ECG Changes: ST elevation ≥ 2mm in ≥ 1 lead from V1-V3 with typical morphology, with an associated RBBB pattern.
2. Imaging, including angiography shows no underlying structural or coronary disease.
3. Stress tests: Symptoms and ECG findings are not usually reproducible with exercise; interestingly, ST elevation normalises with increase in heart rate on exertion4 .
Criteria for the diagnosis of Brugada syndrome have been established as follows5 :
Major criteria
• Presence of ECG marker in structurally normal hearts
• Appearance of ECG marker after administration of sodium channel blockers
Minor criteria
• Family history of sudden cardiac death
• Syncope of unknown origin
• Documented ventricular tachycardia/fibrillation
• Genetic mutation of ion channels
• Positive programmed electrocardiostimulation test on VT / VF
Management
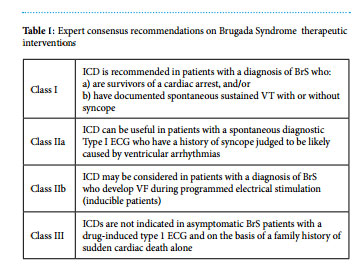
Implantable cardioverter-defibrillator implantation is the mainstay of treatment. Expert consensus recommendations have recently been published by Priori SG et al4 .
Sudden Cardiac Death
With an annual incidence of 0.1-0.2% , sudden cardiac death claims seven million lives per year worldwide. Males are affected three times as much, with a peak incidence between 45 and 75 years. The commonest cause is coronary artery disease, followed by cardiomyopathies and channelopathies. The most common cardiomyopathies are dilated, hypertrophic, arrythmogenic right ventricular, restrictive and left ventricular non-compaction. Channelopathies include Brugada syndrome, long and short QT syndromes, idiopathic VF and short-coupled variant of Torsades de Pointes6 .
Conclusion
The educated eye should look out for the Brugada sign in every electrocardiogram, in order to diagnose and appropriately manage these patients. In addition, the chain of survival following cardiac arrest should be well known to all.

Abbreviations
- BrS – Brugada Syndrome
- ECG – Electrocardiogram
- ICD – Implantable cardioverter-defibrillator
- RBBB – Right bundle branch block
- SCD – Sudden cardiac death
- VT – Ventricular tachycardia
- VF – Ventricular fibrillation
References
1. Ramon Brugada Senior Foundation. brugda syndrome. Available from: www.brugada.org
2. Dizon JM. Brugada Syndrome Clinical Presentation. Available from: http:// emedicine.com
3. Wilde AA, Antzelevitch C, Borggrefe M, Brugada J, Brugada R, Brugada P et al. Proposed diagnostic criteria for the Brugada syndrome: consensus report. Circulation 2002 Nov 5;106(19):2514-9. Available from: PubMed PMID: 12417552.
4. Priori SG, Wilde AA, Horie M, Cho Y, Behr ER, Berul C et al. HRS/EHRA/ APHRS Expert Consensus Statement on the Diagnosis and Management of Patients with Inherited Primary Arrhythmia Syndromes: Document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm 2013 Dec;10(12):1932-63. Available from: doi: 10.1016/j.hrthm.2013.05.014.
5. Gussak I, Bjerregaard P, Hammill SC. Clinical diagnosis and risk stratification in patients with Brugada syndrome. J Am Coll Cardiol 2001 May;37(6):1635-8. Available from: PubMed PMID: 11345377.

