Modern Surgical Techniques for the Treatment of Haemorrhoids
by James Vassallo & Noel Cassar
Abstract
It is said that 50% of people have haemorrhoids, and that the rest are liars. While this is definitely an overestimation, this condition is very common, but not always symptomatic. The fundamental principles of management are the exclusion of possible co-existent disease, and treatment tailored to the patient’s condition and haemorrhoidal grade. There have been recent advances in surgical therapeutic options and a comparison with the established methods is outlined in the following article.
Haemorrhoids are abnormally enlarged anal cushions containing arteriovenous anastomosis, traditionally described as occurring in the 3, 7, and 11 o’clock positions.1 The vascular supply is from branches of the superior rectal artery, which are drained by veins (internal venous plexus) emptying into the superior rectal vein. Internal haemorrhoids, which originate from above the dentate line of the anal canal, occur when these anal cushions are dragged down the canal, and are commonly classified by the Goligher grading system2 (table 1).
|
Grade 1 |
Do not prolapse but may bleed |
|
Grade 2 |
Prolapse on straining and reduce spontaneously |
|
Grade 3 |
Prolapse on straining and have to be manually reduced |
|
Grade 4 |
Permanently prolapsed; irreducible |
Table 1: The Goligher grading system for haemorrhoidal prolapse.
External haemorrhoids are those that originate from varicosities of veins (external venous plexus) draining the territory of the inferior rectal artery, and they occur distal to the dentate line. This article is mainly concerned with internal haemorrhoids though in reality they may co-exist.
Aetiological factors for the development of haemorrhoids are primarily increasing age, passage of hard stool, and raised intra-abdominal pressure. Hence risk factors include a low-fibre diet, prolonged defeacation, and pregnancy. Portal hypertension and anorectal varices are unrelated to haemorrhoids.3
The condition can occur at any age. The prevalence in the general population is in the range of around 4.4-24.5%, with a lifetime prevalence of symptomatic haemorrhoids in people over 50 years being around 50%.4,5 The reluctance to seek medical treatment and lack of symptoms may contribute to an under-estimation of the prevalence.
Haemorrhoids most commonly present with painless rectal bleeding on defeacation.6 This occurs when the passage of stool (usually when particularly hard) damages the mucosa and fresh (arterial) bleeding occurs from the underlying vessels; the blood is bright red and found on the surface of stool and toilet paper. The surface of the lavatory may be stained with blood as well. Other complaints include prolapsed piles, peri-anal itching, tenesmus, mucous discharge, soiling, and/or skin tags. Haemorrhoids can cause pain due to sphincter spasm when they prolapse, the pain being relieved when they return inside the anal canal. If the muscle spasm interferes with haemorrhoidal blood flow an acute haemorrhoidal crisis can result.7 If venous outflow is obstructed strangulation can occur, and if not reduced within 1 or 2 hours thrombosis will follow. If the constriction is tight enough to block the arterial supply, ulceration and gangrene of such haemorrhoid may result. These crises are particularly common in late pregnancy and post-partum, and are usually treated conservatively with ice packs, stool softeners and local analgaesia with resolution usually occurring within 7-14 days.1 Incision and drainage of the thrombosis with/without urgent haemorrhoidectomy is an alternative approach and is mostly considered if the patient presents within 3-4 days from onset; it is safe even in pregnancy.9
Given a characteristic history of haemorrhoids, the clinician should examine the patient to determine the presence and grade of haemorrhoids. It is fundamental that other anal and colorectal conditions are excluded before attributing all the symptoms to haemorrhoids, especially in the older patients.10 In a survey by Koning & Loffeld, 55% of patients with haemorrhoids with a mean age of 67 years Hence, haemorrhoids presenting with rectal bleeding, especially beyond 40 years, should be referred to a colorectal specialist and investigated adequately. The anus is examined using an anoscope and flexible sigmoidoscopy performed to exclude sigmoidorectal pathology since this is the site of approximately 65% of colorectal tumours. Colonoscopy is usually reserved for cases with a suspicious history and/or unidentified anal source of bleeding.7 Figure 1 proposes an algorithm for the management of haemorrhoids.
The symptoms and grade of the haemorrhoids guides treatment choice.6,7 It should be noted that grade and symptoms of haemorrhoids are not significantly correlated.8 Haemorrhoids should be treated only if they interfere with the health-related quality of life (HRQoL) of the patient; grade alone shouldn’t determine treatment choice.
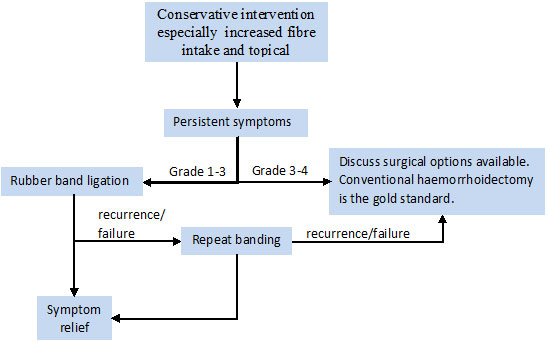
Figure 1: Algorithm for management of haemorrhoids.
The single most important conservative intervention is increasing the daily fibre intake to >25grams/day via the diet with/without fibre supplements.5 Together with increasing liquid intake, minimizing time on the toilet, and evacuating soon after feeling the urge, these interventions are aimed at minimising constipation and straining. Bathing in warm water (approximately 40oC) has a soothing effect on anal discomfort; sitz bathing may be unwise since the setup may promote further venous congestion. Venotonic agents, such as diosmin (a flavonoid), used in addition to the above measures may improve the outcome of conservative treatment;6,11 venotonic injection at the haemorroidal site is also possible but has a poor outcome.5 For quick relief of symptoms topical agents containing local anaesthetics, steroids, astringents, and/or antiseptics may be satisfactory but prolonged application may induce allergy and maceration.
Conservative management is sufficient to improve or resolve symptoms in many patients. Overall, fibre supplementation can decrease severity of symptoms by a mean of around 50% in patients with grade 1-3 haemorrhoids and hence can be sufficient to render the haemorrhoids non-significant as regards to the HRQoL.12 In fact fibre supplementation is as effective as sclerotherapy.6 Persistent symptoms beyond 1 month of conservative therapy should prompt consideration regarding more invasive treatment.7
Non-operative procedures are usually considered first for persistent grade 1-3 haemorrhoids. The gold-standard is rubber band ligation.13 It has the lowest recurrence rate at 12 months compared to sclerotherapy and infrared photocoagulation. It is recommended as the first line treatment for grade 1 and 2 haemorrhoids.14 This technique was originally devised in 1958 by Blaisedell, improved by Barron in 1963, and has undergone significant modifications over time, such as the application of up to 3 bands per session as opposed to one.6,15 No bowel preparation is required. The equipment typically employs suction, as in the McGown ligator, to draw up the pedicel of the haemorrhoid 1cm or more above the dentate line, and then fires a band which strangulates the blood supply of the haemorrhoid causing it to shrivel off.5 The patient should be warned of anticipated rectal bleeding at 5-14 days after the procedure. Correct band positioning is essential to minimise post-ligation pain; indeed such a procedure is meant to be performed without local anaesthesia as an outpatient procedure. Mild/moderate pain occurs in roughly 30% of cases. Success rate is around 75% and the complication rate stands at 0.7%. Recurrence at 4-5 years is around 70% but repeating the procedure is usually sufficient;5 only 10% of cases would require eventual haemorrhoidectomy. A significant bleeding tendency, and treatment with warfarin or heparin are absolute contra-indications for banding. Anti-platelet agents, such as aspirin, should be withdrawn for a week before and after the procedure. When banding is contra-indicated, other non-operative approaches can be offered. The main alternatives are sclerotherapy, bipolar diathermy, infrared photocoagulation, cryotherapy, and venotonic injection, although these have variable success rates.
For symptomatic grade 3-4 haemorrhoids and haemorrhoids resistant to non-operative procedures, a surgical approach can be adopted. This is required in only 5-10% of patients.5 Many techniques for haemorrhoid excision have been described but the most commonly used are the open Milligan-Morgan haemorhoidectomy (MMH) and the closed Ferguson haemorrhoidectomy (FH), which differ in whether the skin wounds are left open to heal by secondary intention or else are sutured primarily in the case of the Ferguson method. MMH may be overall better than FH particularly as regards complication rate.5 The major considerations accompanying open haemorrhoidectomy are the significant post-operative pain and the protracted recovery time (a minimum of 4 weeks with the MMH). Haemorrhoidectomy may be carried out as a day-case procedure.15 A post-operative plan for pain relief devised in alliance with the patient is very important for better recovery. Other possible short-term complications include urinary retention, bleeding, and infection; long-term concerns include anal stenosis, feacal incontinence, anal fissure, and fistula-in-ano.4
Circular stapled haemorrhoidopexy is another, recently introduced, operative technique for haemorrhoids.4 This technique is also known as ‘procedure for prolapse and haemorrhoids’ (PPH) or stapled anopexy/mucosectomy/prolapsectomy. PPH was introduced by Longo A. in 1998 and employs a circular stapling device which removes mucosa and submucosa circumferentially 2-3cm above the dentate line, anastomosing the proximal and distal edges, interrupting the blood supply to the remnant haemorrhoidal tissue.16 PPH is significantly less painful and allows quicker recovery than MMH, but the recurrence rate may be higher in the long run. One study showed the recurrence rate of PPH vs MMH to be 5.7% vs 1% at 1 year and 8.5% vs 1.5% overall.5 A recent meta-analysis showed that although the short term benefits of stapled haemorrhoidectomy may be better, the recurrence rate is significantly higher.17
A promising procedure is the application of Doppler-guided haemorrhoidal artery ligation (HAL), first described by Morinaga K et al in 1995.18 This technique can be performed under sedation and/or local anaesthesia and involves a proctoscope with a Doppler transducer integrated in the probe. This allows sequential identification of the position and depth of superior rectal arterial branches (usually 5-7 are found at one level) which are then selectively ligated 2-3cm above the dentate line at two levels 1-1.5cm apart by absorbable sutures via a lateral ligation window within the scope.16 The interference with the blood supply suppresses the bleeding and volume of the haemorrhoids and symptomatic relief is usually evident within 6-8 weeks. Several studies have found this technique to give good results for grade 2 and 3 haemorrhoids,19,20 but randomised clinical trials and long term follow up are awaited to compare this technique with the open method. In 2005 the technique was improved with adjunctive recto-anal repair (HAL-RAR). Following ligation, HAL-RAR includes pulling up the prolapsed masses by means of running sutures and hence folding them into the anal canal. Hence symptom relief for grade 3-4 haemorrhoids can be achieved as well.16
Other techniques for haemorrhoidectomy include the application of the LigaSureTM System, Harmonic Scalpel, and laser surgery.5 Dissection and coagulation are achieved via the application of pressure with graded electrical energy or fine oscillatory motion with the LigaSureTM and Harmonic Scalpel instruments respectively, providing precision, a relatively bloodless field, and minimal collateral tissue damage. These methods are not widely used yet but some reports show initial positive results.21-22
Conclusion
Conventional haemorrhoidectomy is the gold standard operation against which other haemorrhoidal procedures should be compared. Nonetheless, it has its own post-operative morbidity, including pain, bleeding and infection. This has led to the application of more recent techniques to improve the treatment of this very common disease. General practitioners and colorectal surgeons have to be familiar with these novel treatment options so as to be able to guide their patients appropriately.
References
- Burkitt HG, Quick CRG, Reed JB. Anal and perianal disorders.
- Goligher JC. Surgery of the anus, rectum and colon. 5th London: Bailiere Tindall, 1984.
- Hosking SW, Smart HL, Johnson AG, Triger DR. Anorectal varices, haemorrhoids, and portal hypertension. Lancet 1989; 1(8634):349-52.
- NICE technology appraisal guidance 128. Stapled haemorrhoidopexy for the treatment of haemorrhoids. 2007.
- Sneider Eb, Maykel JA. Diagnosis and management of symptomatic hemorrhoids. Surg Clin N Am 2010; 90:17-32.
- Acheson AG, Scholefield JH. Management of haemorrhoids. BMJ 2008; 336:380-3.
- Thornton SC. Hemorrhoids [internet]. 2010 [updated 2010 Mar 16; cited 2010 Sep 5]. Available from:
- Gerjy R, Lindhoff-Larson A, Nyström PO. Grade of prolapse and symptoms of haemorrhoids are poorly correlated: result of a classification algorithm in 270 patients. Colorectal Disease 2008; 10(7):694-700.
- Saleeby RG, Rosen L, Stasik JJ et al. Hemorrhoidectomy during pregnancy: risk or relief? Dis Colon Rectum1991; 34(3):260-1.
- Koning MV,Loffeld RJ. A survey of abnormalities in the colon and rectum in patients with haemorrhoids. BMC Gastroenterol 2010; 10:74.
- Ho YH,Tan M, Seow-Choen F. Micronized purified flavonidic fraction compared favorably with rubber band ligation and fiber alone in the management of bleeding hemorrhoids: randomized controlled trial. Dis Colon Rectum 2000; 43(1):66-9.
- Alonso-Coello R Guyatt G, Heels-Ansdell D et al. Laxatives for the treatment of hemorrhoids. Cochrane Database Syst Rev. 2005; 4:CD004649.
- MacRae HM, McLeod RS. Comparison of hemorrhoidal treatment A meta-analysis. Dis Colon Rectum 1995; 38:687-94.
- Shanmugam V, Campbell KL, Loudon MA et al. Rubber band ligation versus excisional haemorrhoidectomy for haemorrhoids. Cochrane Database of Syst Rev. 2005; 1:CD005034.
- Barron J. Office ligation treatment of hemorrhoids. Dis Colon Rectum 1963; 6:109-13.
- Evans CFM, Hyder SA, Middleton SB. Modern surgical management of haemorrhoids. Pelviperineology 2008; 27:139-42.
- Giordano P, Gravante G, Sorge R, Ovens L, Nastro P. Long-term outcomes of stapled hemorrhoidopexy vs conventional hemorrhoidectomy: a meta-analysis of randomized controlled trials. Arch Surg 2009; 144(3):266-72.
- Morinaga K, Hasuda K, Ikeda T. A novel therapy for internal hemorrhoids: ligation of hemorrhoidal artery with a newly devised instrument (Moricorn) in conjunction with a Doppler flowmeter. Am J Gastroenterol 1995; 90:610-
- Felice G,Privitera A, Ellul E, Klaumann M. Doppler-guided hemorrhoidal artery ligation: an alternative to hemorrhoidectomy. Dis Colon Rectum 2005; 48 (11):2090-3.
- Wilkerson PM, Strbac M, Reece-Smith H, Middelton SB. Doppler-guided haemorrhoidal artery ligation: long-term outcome and patient satisfaction. Colorectal Dis 2009; 11(4):394-400.
- Tan EK,Cornish J, Darzi AW, Papagrigoriadis S, Tekkis PP. Meta-analysis of short-term outcomes of randomized controlled trials of LigaSure vs conventional hemorrhoidectomy. Arch Surg 2007; 142(12):1209-18.
Tan KY, Zin T, Sim HL et al. Randomized clinical trial comparing LigaSure haemorrhoidectomy with open diathermy haemorrhoidectomy. Tech Coloproctol 2008; 12(2):93-7.

