Recent Advances in Cardiovascular Imaging Part II

Saint James
Imaging, namely cardiac computed tomography (CCT) and cardiovascular magnetic resonance imaging (CMR), in the structural and functional assessment of the cardiovascular system. Although Computed Tomography and Magnetic resonance have been in clinical use over the last 40 years, it is only through more recent advances in technology that imaging of a constantly moving structure like the heart has become possible with sufficient acquisition speed and spatial resolution. Now that this technology has become an important component of mainstream cardiology, it is imperative that all medical professionals understand the role and capabilities of CCT and CMR in order for patients to benefit from the exquisite diagnostic potential of this non-invasive technology.
Cardiac Computed Tomography
The turning point which led to cardiac imaging is the development of helical CT in 1997. Further improvements in gantry rotation speed, image processing techniques and detector size have enabled rapid acquisition of images of coronary arteries with sufficiently high spatial resolution (sub-millimetre) and low motion artefact to be able to resolve presence of atherosclerotic plaque and luminal stenosis. CCT differs from imaging of other organs in that acquisition is gated to the patient’s ECG in order to acquire images during phases when cardiac motion is minimal, and in order to reconstruct images according to the phase of the cardiac cycle. The patient is also instructed to breath-hold for a few seconds to minimise respiratory motion.
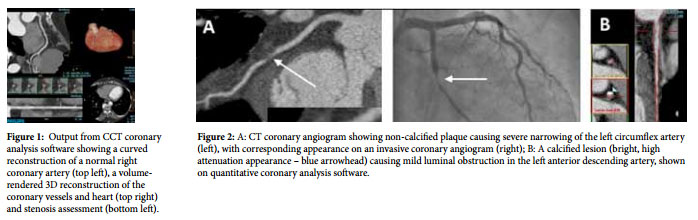
The spatial resolution of modern CCT rivals that obtained in conventional invasive coronary angiography. However, the main advantage of CCT is the ability to visualise and characterise atherosclerotic plaque, even when it does not cause significant luminal obstruction. Invasive angiography acquired during the intracoronary injection of radio-opaque contrast media, on the other hand, only reveals the luminal calibre, and fails to visualise non-calcified plaque which does not impinge on the lumen. Yet the presence of such plaque is known to be a precursor of acute coronary syndromes. The other obvious advantages of CCT are its non-invasive nature, avoidance of potentially life-threatening complications, and drastically reduced in-patient stay (the whole procedure rarely takes more than 20 minutes).
The main use of CCT is in the detection of coronary artery disease (CAD) in patients with chest pain deemed to be at low to intermediate risk of having significant CAD (figures 1 and 2). The rationale behind this is two-fold: one would like to benefit from the CCT’s high negative predictive value for CAD in this subgroup and, secondly, the careful selection of patients who are unlikely to need further downstream exposure to radiation due to invasive angiography and percutaneous revascularisation. CCT coronary angiography is also useful in the presence of equivocal results from stress ECG or functional imaging (stress echocardiography or nuclear perfusion scans) in patients with suspected angina.
CCT has two other important roles. The first is the quantification of coronary calcium for the purpose of cardiovascular risk stratification in asymptomatic patients with intermediate cardiovascular risk (based on presence of cardiovascular risk factors and use of risk scores such as Framingham risk score or Heart Score) or in patients with a positive family history of premature ischaemic heart disease. The presence of coronary calcification is practically synonymous with the presence of atherosclerotic plaque, and can be quantified by scores based on the degree of attenuation and area of the lesion (Agatston score) (figure 3) An age- and gender-based percentile can be derived for the patient based on nomograms derived from population studies. The resulting refined risk stratification may aid decisions on starting primary prevention medication, based on the amount of calcification. A CT calcium score may tip the risk-benefit ratio one way or the other if you are not sure whether your asymptomatic patient may derive a net benefit from statins or aspirin for primary prevention. In addition, a CT calcium score may prove useful as a gatekeeper in patients with non-acute chest pain with low probability of CAD – while the absence of calcium implies an excellent prognosis in this subgroup of patients and obviate the need for further imaging, the presence of any degree of calcium may prompt the imager to proceed with CT angiography.
The second major role of CT is in the assessment of heart chambers, valves, pericardium, congenital heart disease and major blood vessels (figure 4). Such scans are usually indicated when information cannot be obtained from non-radiation techniques like echocardiography or CMR (commonly due to poor echocardiographic windows and/or presence of metal implants – like cardiac pacemakers – which preclude a CMR). Although this involves considerably more radiation, X-rays can be emitted throughout the cardiac cycle to obtain systolic and diastolic images, allowing calculation of ejection fraction and stroke volume (figure 5)
Like invasive angiography, CCT entails exposure to ionising radiation. However, careful patient selection, pre-procedural beta-blockers to reduce the patient’s heart rate, use of special acquisition techniques, and other technological improvements have dramatically reduced the radiation dose. With a state-ofthe-art scanner in a well selected patient, one should expect an effective dose of 3 mSv, which compares favourably with invasive diagnostic coronary angiography (2-7 mSv) and nuclear perfusion imaging (6-15 mSv).

Cardiovascular Magnetic
Resonance Imaging Like CCT, CMR is based on well-established technology which had to await further technological innovations before becoming feasible for cardiac imaging. CMR is a non-invasive technique which does not utilise ionising radiation. Magnetic resonance imaging is based on the behaviour of hydrogen nuclei, mostly within water molecules, when placed in a magnetic field generated by a superconductor magnet. Excitation by radiofrequency signals temporarily deflects magnetisation of these hydrogen nuclei. While this altered magnetisation returns to its equilibrium state, a current is induced in a receiver coil, generating the magnetic resonance signal which is used to construct an image.
The beauty of CMR is that excellent intrinsic contrast, without administration of exogenous contrast, is generated between tissues that have different chemical composition. However, exogenous contrast like gadolinium is also administered to produce high quality angiograms (figure 6) and to determine the cause of heart muscle disease (figure 7). By being able to provide functional information (blood flow velocity and stroke volume), CMR is a truly “one-stop shop” for addressing all cardiac problems. The other advantage of CMR over echocardiography is the independence of image quality from body habitus and unusual anatomy; indeed imaging planes can be prescribed in any direction and are not limited by the factors that impede ultrasound transmission through body tissues.
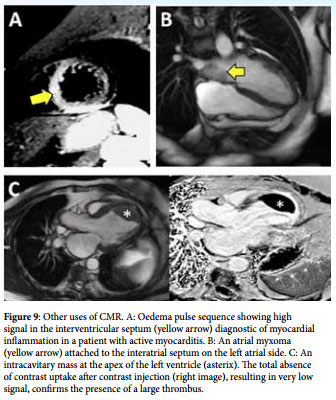
CMR provides a wealth of structural and functional information, ranging from myocardial abnormalities, valve disease, aortic pathology, cardiac tumours and congenital heart defects (figures 8 and 9). Perfusion CMR is a technique for detecting cardiac ischaemia; imaging is carried out during gadolinium infusion at baseline and during vasodilator stress with adenosine. Hypoperfused myocardium manifests as low signal areas during stress imaging, which are not visible on the resting images (figure 8D). A common indication is to assess the cause of heart failure, and to assess the viability of heart muscle – in patients with myocardial infarction – to guide revascularisation of CAD (figure 7). The severity of valvular stenosis and regurgitation can be confirmed with CMR (avoiding the need for invasive pressure measurements) by measuring transvalvular velocities and regurgitant volumes. Another important role of CMR is in the noninvasive detection of cardiac iron overload in patients who require lifelong blood transfusions, as in thalassaemia major, prompting timely administration of life-saving chelation therapy. Finally, since CMR does not involve radiation, it is ideal for lifelong follow-up of patients with inherited cardiovascular conditions and operated congenital heart disease patients, most commonly patients with repaired tetralogy of Fallot and patients with Marfan’s syndrome and dilated aortas.
CMR does have some important limitations. Patients with certain metallic implants cannot enter the scanner due to the deleterious effect of the strong magnetic field. Cardiac pacemakers are a particularly common problem, though most companies now produce MRI-conditional pacemakers which can be placed in a scanner provided certain precautions are taken. Another problem is the duration of the scan – patients are expected to lie still for about 45 minutes on average. Difficulties in suspending respiration and arrhythmias can affect image quality. Claustrophobia is a real problem, though sedation and use of wide-bore scanner systems have proved to be helpful. Large patients may not fit in the scanner. Of note, patients who cannot have an MRI because of these reasons can be considered for CT scanning, as discussed above. Finally, gadolinium contrast is contra-indicated in patients with severe renal dysfunction although, fortunately, there are non-contrast alternatives available in most situations.
Cardiac imaging has changed modern cardiology practice, and continues to do so as technology improves. The limitations of echocardiography and the need for invasive imaging have been eliminated for a large subgroup of our patient population. The techniques described are well-grounded in science and practical experience. In these two articles I have addressed the role and indications of these new cardiac imaging technologies, most of which are already available in Malta.