Wellbutrin XR

|
|||
| Based on references 1, 4-8 | |||
 |
|||
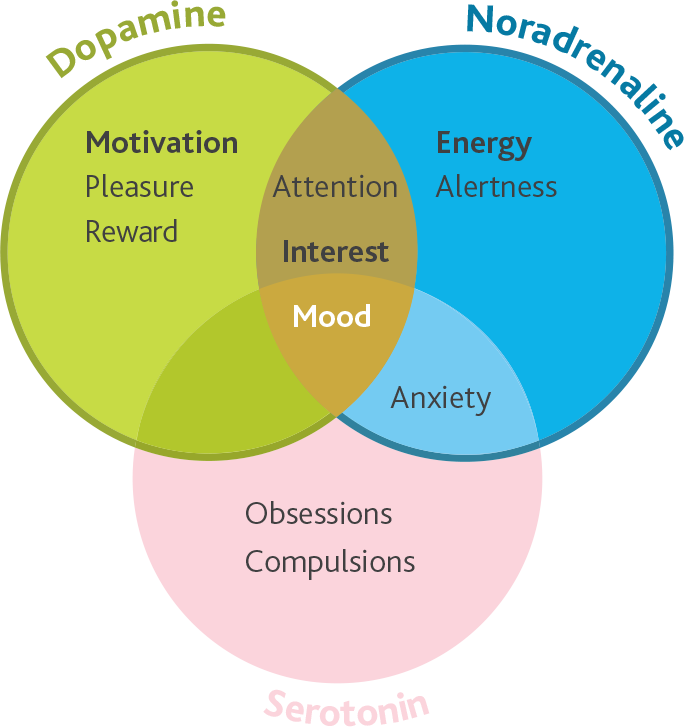
Wellbutrin XR is indicated for use in adults over 18 years of age for the treatment of major depressive episodes |
|||
 |
|||
 |
|||
|
|||
 |
|||
| WELLBUTRIN XR is contraindicated in patients:9 | |||
|
|||
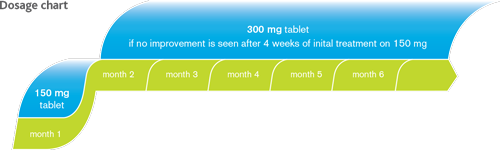
| Concomitant use of WELLBUTRIN XR and monoamine oxidase inhibitors (MAOIs) is contraindicated. At least 14 days should elapse between discontinuation of irreversible MAOIs and initiation of treatment with WELLBUTRIN XR. For reversible MAOIs, a 24 hour period is sufficient.9 The recommended dose of modified release bupropion tablets should not be exceeded, since bupropion is associated with a dose-related risk of seizure. The overall incidence of seizure with modified release bupropion tablets in clinical trials at doses up to 450 mg/day was approximately 0.1%.9 Very common side effects with Wellbutrin XR use could be insomnia, headache, dry mouth, gastrointestinal disturbance including nausea and vomiting.9 In order to ensure that this product information reflects the most up-to-date clinical and post-marketing surveillance data, please always refer to the latest Summary of Product Characteristics (SPC) which is available from GlaxoSmithKline (Malta) |
|||
| REPORTING ADVERSE EVENTS (AEs): If you become aware of any AEs, medication errors and/or use during pregnancy in association with GSK products, please report the event promptly to: GSK (Malta) Limited, 1, De la Cruz Avenue, Qormi QRM 2458, Malta (Tel: +356 21238131) Alternatively, any suspected AEs and medication errors can also be reported via the national Adverse Drug Reactions (ADRs) reporting system: |
|||
| Wellbutrin XR should not be used together with other Bupropion containing medicinal products. Wellbutrin XR tablets should be swallowed whole and not crushed or chewed. | |||
| References | |||
|
|||
| Job No: MLT_GIB/BHC/0001/16 Date of preparation: March 2016 |
|||
|